 Zooplankton of the Great Lakes
Zooplankton of the Great Lakes
Zooplankton of the Great Lakes Home Page
Rotifer: Polyarthra vulgaris
Domain: Eukarya
Kingdom: Animalia
Phylum: Rotifera
Class: Monogonata
Order: Ploimida
Family: Synchaetidae
Genus: Polyarthra
Specific epithet: vulgaris
Anatomy
P. vulgaris is an aloricate rotifer, with 4 sets of blade-like projections or paddles (Gilbert 1985). A ciliated corona surrounds the mouth leading to the gullet, trophi (jaws), esophagus, stomach, and intestine, and the anus on the ventral surface. The cilia surrounding the corona are used in feeding and locomotion. Females (most common in collections) possess a large ovary consisting of a germarium and vitellarium. The oviduct empties into the cloaca. The male reproductive system contains a testis and sperm duct. The tactile sense organs consist of the two lateral antennae and dorsal antenna and a cephalic retrocerebral organ (Edmondson, 1959).
The paddles are attached to strong, striated muscles within the body by integumentary hooks. When the muscles contract, they pull the hooks downwards and cause the paddles to elevate. As the rotifer lacks any protective outer covering, it is the rapid movement of the paddles that allows it to escape predation (Gilbert, 1985).
Distribution
P. vulgaris is common in the epilimnion of lakes worldwide (Edmondson, 1959). It is more common in the limnetic zone than the littoral zone (Jose de Paggi, 1995)., and most populations are limited by algae availability and predation (Foran and King, 1982). Common predators include protozoans, Hydra, cladocerans, copepods, Chaoborus , mysids, and predatory rotifers (Williamson, 1983). Movements are often dictated by prey organisms such as flagellated green algae (Salonen and Rosenburg, 2000.
Reproduction and Life History
P.vulgaris contains two types of reproducing females. Amictic females are parthenogenetic, and mitcic females produce haploid males from unfertilized eggs and diploid females from fertilized eggs. The fertilized eggs are also called diapause eggs or resting eggs, and can persist for extended periods of time without hatching. These develop into amictic females which differ morphologically from the parent generation, often by having an increased lipid content, a reduced digestive tract, and by producing a single large egg only hours after hatching (compared to the normal 6 day generation time). While not all the environmental triggers for resting egg production have been determined, it is possible that this strategy allows for quick parthenogenetic population growth while environmental conditions are still too uncertain to invest resources in bisexual reproduction (Gilbert and Schroder, 2004).
Feeding Ecology
P. vulgaris feeds mainly on small organisms such as bacteria, single-celled green algae, diatoms (Bogden and Gilbert, 1987), and chrysophytes (Devetter, 1998). Feeding is probably accomplished through pharyngeal expansion and suction (Bogden and Gilbert, 1987. Fecundity of P. vulgaris populations shows a positive relationship with the amount of chlorophyll-a present in an aquatic system, indicating their nutritional dependence on small photosynthetic organisms (Devetter and Sed'a, 2003). In some cases, particularly during unstable hydrological conditions, P. vulgaris and other rotifer species are the dominant aquatic grazers and responsible for the majority of carbon transactions in the planktonic food web (Keckeis et. al. 2003).
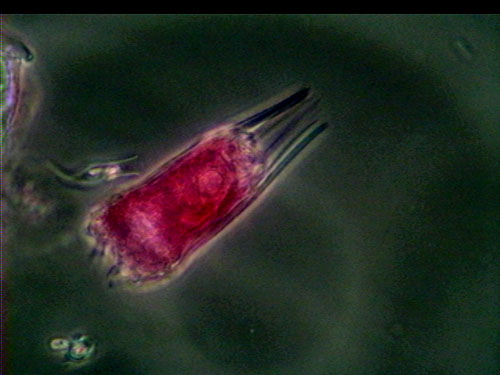
Figure 1: P. vulgaris with "paddles" streaming behind the body. At the anterior (left) end, trophi and cilia are visable.
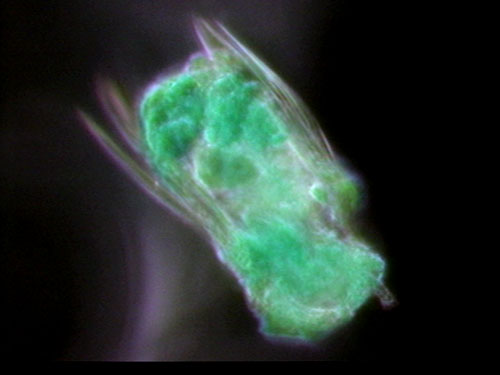
Figure 2: P. vulgaris, green hue added through image editing.
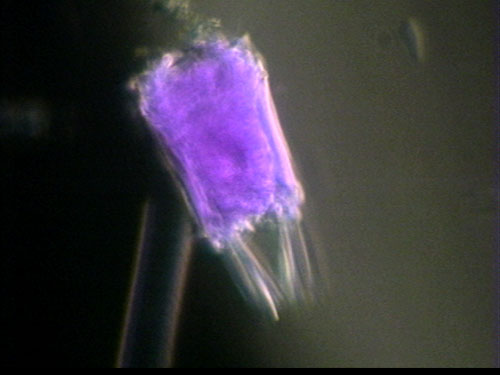
Figure 3: P. vulgaris, violet hue added through image editing.
Links
Gallery of Rotifers Some general rotifer biology and lots of pictures (non-Polyarthra spp.)
Introduction to the Roitfera Pictures, information, and links to lots of other resources.
Grow Your Own! Information about culturing rotifers.
Works Cited
Bogdan, KG., and JJ. Gilbert. 1987. Quantitative comparison of food niches in some freshwater zooplankton; a multi-tracer cell approach. Oecologia (72): 331-340.
Devetter, M. 1998. Influence of environmental factors on the rotifer assemblage in an artificial lake. Hydrobiologia (387/388): 171-178.
Devetter, M., and J. Sed'a. 2003. Rotifer fecundity in relation to components of a microbial food web in a eutrophic reservoir. Hydrobiologia (504): 167-175.
Edmondson, WT, editor. 1959. Freshwater biology: 2nd edition. New York: John Wiley and Sons, Inc.
Gilbert, JJ. 1985. Escape response of the rotifer Polyarthra: a high-speed cinematographic analysis. Oecologia (66): 322-331.
Gilbert, JJ., and T. Schroder. 2004. Rotifers from diapausing fertilized eggs: unique features and emergence. Limnology and Oceanography ( 49): 1341-1354.
Foran, JA., and RH. King. 1982. A regression analysis of the summer population dynamics of Polyarthra vulgaris in a northern Michigan bog lake. Hydrobiologia (94): 237-246.
Jose de Paggi, S. 1995. Vertical distribution and diel migration of rotifers in a Parana River floodplain lake. Hydrobiologia (310): 87-94.
Keckeis, S., Baranyi, C., Hein, T., Holarek, C., Reidler, P., and F. Schiemer. 2003. The significance of zooplankton grazing in a floodplain system of the River Danube. Journal of Plankton Research (25): 243-253.
Salonen, K., and M. Rosenburg. 2000. Advantages from diel vertical migration can explain the dominance of Gonyostomum semen (Raphidopphyceae) in a small, steeply-stratified humic lake. Journal of Plankton Research (22): 1841-1853.
Williamson, CE. 1983. Invertebrate predation on planktonic rotifers. Hydrobiologia (104): 385-396.